For many people, having overly large breasts is not just an aesthetic concern—it’s a daily physical and emotional burden. Chronic… Read More
Breast Augmentation
Home/Breast Surgery Procedures/Breast Augmentation
Home/Breast Surgery Procedures/Breast Augmentation
Breast augmentation (also referred to as breast enlargement, breast enhancement or augmentation mammoplasty) is the surgical procedure that enlarges a woman’s breast size and improves its shape. It has been among the most popular cosmetic surgical procedures for decades and is associated with an extremely high satisfaction rate. Surgery involves the insertion of silicone or saline implants into the appropriate location deep within the breasts in order to increase their size.
The concept and procedure sound rather simple and straightforward but this is definitely not the case. In order to obtain the most ideal results, many variables need to be considered and decisions made prior to surgery. The goal of any breast augmentation procedure is to add volume and improve the shape of a woman’s breasts while keeping the tell-tale signs of surgery to an absolute minimum. To achieve this goal, the plastic surgeon must have an exceptional eye for aesthetics, an understanding of each individual’s tissue characteristics, an appreciation for the three dimensional effects of the implants on the tissues, consideration for the changes over time and superior surgical skills to implement these strategies. In addition, he/she must have the ability to understand and interpret the patient’s goals and needs and then translate that into outstanding results.
It is truly an art and science!
With these abilities and meticulous attention to all details and variables, Scottsdale plastic surgeon Dr. Steven H. Turkeltaub has consistently endeavored to ensure that the results he creates for his patients are exactly what they had desired. (See blog: Breast Augmentation Fail or When Not to Disclose One’s Assets to see an example of what does happen when surgical planning, precision and skill are not what they should be.)
I went to Dr. T. for a breast augmentation on recommendations of two of my friends who were very happy with their results. He listened to what I wanted, spent alot of time with me and gave me all the information that I needed to make my decision. I really liked him and his recommendations and decided to have him do my surgery. My breasts are perfect – exactly what I wanted! I am so happy now. I can definitely recommend him to other friends for their surgery.
Nikki Johnson
September 16, 2017

While breast implants are most commonly associated with achieving a larger breast size, this is by no means the sole enhancement and/or purpose they can provide. In many cases, implants can be an excellent option for improving the shape or symmetry of the breasts, restoring youthful volume following pregnancy or dramatic weight loss, addressing congenital issues (like Poland’s syndrome) or even reconstructing one or both breasts following mastectomy—with or without necessarily increasing one’s natural breast size appreciably.
Based on your unique needs and goals, Dr. Turkeltaub can help you select the most appropriate implant type, size and shape for the breast aesthetic you desire.
Beyond the immediate goal of enhancing breast size and volume, breast implants can also profoundly influence confidence, comfort and overall quality of life. For the right patient, the breast augmentation procedure can facilitate the following improvements:
In these ways, breast augmentation can be a pathway towards improved confidence, renewed comfort in a patient’s own skin and greater long-term satisfaction with one’s appearance. These influences can generally contribute to a happier, more positive well-being.
Breast implants can be an excellent solution for a diverse range of women seeking to enhance their breast appearance but they are not exclusively reserved for those desiring a larger cup size. Ideal candidates often include:
In addition to physical considerations, good candidates for breast implants should be in generally good health, non-smokers (or willing to quit), and possess realistic expectations regarding the results of surgery. Emotional readiness is equally important, as breast augmentation is a personal decision that should be made for one’s own satisfaction rather than to meet external pressures or ideals.
Dr. Turkeltaub takes a personalized approach to every consultation, carefully evaluating your aesthetic goals, anatomical features, and lifestyle to determine the best course of action. Should you decide to proceed with implants, he will guide you in selecting the optimal implant type, size, profile and placement technique to achieve a beautiful and natural-looking enhancement tailored specifically to you.
Breast augmentation is an outpatient procedure that is most often performed under general anesthesia. This approach is very safe and much more comfortable than a “twilight” or semi-awake type of anesthetic. It can be combined with many other cosmetic procedures, such as tummy tucks (as part of a “Mommy Makeover”) and liposuction, for a more extensive and cost effective improvement.
During surgery, the most appropriate implant for the patient will be selected based on the preoperative discussion of all the options and the patient’s goals. Dr. Turkeltaub uses a very precise, meticulous and gentle technique which also helps to minimize both the postoperative discomfort as well as the risk for less than ideal outcomes.
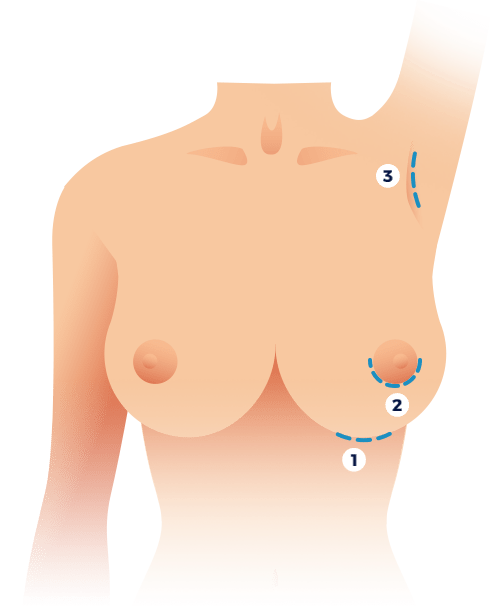
Breast augmentation patients can select to have their implants placed through incisions in one of three different locations. The transumbilical approach, where the implants are inserted through an incision at the belly button, is not considered and for a variety of reasons.
Breast implants can be placed either behind (subpectoral or submuscular) or directly in front of (submammary or pre-pectoral) the pectoralis major muscle, the large chest wall muscle. The appearance and dynamics are different in these two locations as are the consequences of the effects of time.
For most patients, Dr. Turkeltaub recommends that the implants be situated behind the muscle.
In general, if you have a moderate to significant amount of excess skin of your breasts along with drooping you will likely need a breast lift (mastopexy) in combination with your breast augmentation in order to achieve desirable results. Having a breast enlargement alone without removing extra skin or lifting breast tissue in these cases would result in abnormal and bizarre appearing breasts.
Some surgeons, trying to avoid performing a lift in these situations, place extremely large and inappropriately sized implants often above the muscle in a misguided effort to take up the skin slack and avoid the additional incisions associated with the lift. The long term consequences of this can be extremely bad and very challenging to address at a later time.
During your consultation, Dr. Turkeltaub can help you determine if a breast augmentation alone or in combination with a mastopexy would be best for your needs and goals. If you would like to learn more about this option before scheduling a consultation, please feel free to explore our page on breast augmentation with a lift.
A surgical dressing providing compression and maximizing comfort is placed following surgery and remains on for around 2 – 3 days. This is then replaced with a sports type bra that is worn day and night for at least 2 weeks. After that, a supportive bra should be worn on a regular basis, especially if you have relatively large implants, so as to help maintain the breast shape and minimize drooping over time.
Discomfort for the first few days can be mild to moderate though most patients can return to a non-strenuous job or school within 5 – 7 days. It is recommended that you avoid vigorous activities such as sports and aerobics for at least three to four weeks. You may experience muscle tightness and even spasms early on but these resolve fairly quickly.
Initially your breasts will be swollen, firm and may appear larger than you were anticipating. After several weeks, much of the inflammation will subside resulting in a more defined shape and a size closer to what you can expect long term. With time, the skin and muscle (if placed in a submuscular location) will relax resulting in a softer, more natural feel and appearance.
There are many long-term benefits associated with breast enlargement. Included among these are looking better, feeling more attractive and feminine, having greater self-esteem and being able to find clothes that fit and that are more complementary.
The overwhelming majority of breast enhancement patients are extremely pleased with their results.
For more information on the postoperative period after breast augmentation surgery, read Dr. Turkeltaub’s Blog: “Recovering From Breast Augmentation Surgery.”
The visibility and overall appearance of breast augmentation scarring will depend on a variety of elements including incision location, one’s genetic makeup, the incision closure method employed, level of surgeon’s skill, adherence to post-surgical advice, healing response, usage of scar mitigation products and whether one is on a restrictive diet (vegetarian, vegan, etc.) or not. Most commonly, the incision used to place implants will be made in or near the inframammary fold, resulting in a scar that is concealed beneath the breasts. Other scar locations include along the outer border of the lower half of the areola or in the armpit, both of which can result in relatively inconspicuous scarring.
In Dr. Turkeltaub’s experience, breast augmentation patients rarely have any issues with the final appearance of their scars particularly when post-operative instructions are closely followed and recommended scar care products/treatments are utilized.
Breast implants are not guaranteed to be lifetime devices and given enough time, all will ultimately lose their integrity. However, they can last up to 30 years or even more. Silicone implants generally last significantly longer than saline ones.
Being able to determine when one’s implants will need to be replaced or removed (explanted) because they have worn down or lost their integrity is a relevant concern. How you can realize when it is that time is quite different depending on whether you have silicone or saline breast implants.
Ultimately, if there is any concern about the integrity/condition of your breast implants especially if they are getting up there in the years, you should seek the advice of your plastic surgeon. Read more about breast implant durability.
If your body is capable of producing breast milk prior to the placement of your implants, you should still be able to breastfeed after your augmentation procedure, particularly if the periareolar incision technique is not utilized. It is important to understand, however, that some women are not capable of producing a sufficient amount of milk for breastfeeding regardless of whether or not they have breast implants. Therefore, if you have never tried to breastfeed prior to breast augmentation, your ability to do so after receiving breast implants may be uncertain. This fact greatly contributes to the misconception that breast implants impact one’s ability to breastfeed.
Another false belief regarding breastfeeding after breast augmentation is that material from the implants will contaminate the breast milk. In actuality, the cohesive silicone gel or saline solution inside implants should not get into the breast milk or harm milk production glands and/or breast tissues. However, the periareolar approach for implant insertion can lead to some risk of disrupting milk glands. This is another reason why Dr. Turkeltaub recommends inserting breast implants through an inframammary incision for virtually all of his breast augmentation patients.
Ultimately, if you wish to start—or grow—a family after breast augmentation and are hoping to breastfeed, Dr. Turkeltaub suggests that you disclose this information during your consultation. By doing so, your plastic surgeon can more appropriately plan your procedure.
The vast majority of women undergoing regular screening of their breasts for breast cancer will have the standard two image mammogram. A relatively small percentage will use the ultrasound approach. For those women who either have had a breast augmentation or are considering one, it is therefore important to know that the breast implants used in a breast augmentation will impact the quality of routine mammograms to a varying degree. Fortunately, this can usually be easily addressed with very common standard modifications of the study.
Mammograms use very low energy x-rays to penetrate through your tissues in order to radiographically visualize your breasts. Both silicone and saline breast implants partially block penetration by these x-rays making tissue visualization and evaluation on mammograms less clear and more difficult. The effective solution for this typically is having two additional views taken along with some repositioning of the implants and/or breasts. Therefore, it is always important that when you are scheduling your mammogram as well as when you are having it done, you inform the appropriate people that you do have breast implants so proper measures can be taken.
For more information on this, read Dr. Turkeltaub’s blog: “Do Breast Implants Interfere with Mammograms Evaluations?”
The American Society of Plastic Surgeons (ASPS) reports the average plastic surgeon’s fee for a breast augmentation in 2023 is approximately $4,516*. This price is a bit misleading, however, because it refers solely to the plastic surgeon’s fee and does not take into account other related and inherent costs including the specific implants chosen, the surgical venue (hospital or private operating facility) and anesthesia expenses. Additional components that are also not accounted for in this number include such items as medications during recovery and preoperative mammograms. Of course, these numbers will also be affected if a breast lift is performed as well.
To receive a personalized quote for the cost of your breast augmentation, a consultation with Dr. Turkeltaub is necessary. During this meeting, he will listen as you describe the look you wish to attain, help you select the most appropriate implants based on your goals and desires and then formulate a customized surgical approach for you. Once a treatment plan is agreed upon, he can then provide you with a breakdown of all expenses that contribute to the total cost.
If you are considering financial assistance for breast augmentation, a member of our team will be available to introduce you to several healthcare lenders that offer affordable payment plans for almost any budget. The online application process is simple and the decision regarding approval is generally obtained quickly.
*This price is taken from data collected by the American Society of Plastic Surgeons; it does not represent the exact cost of breast augmentation at Dr. Turkeltaub’s practice.
While breast augmentation on its own can produce dramatic and confidence-boosting improvements, many patients choose to further refine their results by combining it with complementary procedures. These additional treatments can address other areas of concern and create a more balanced, harmonious outcome — both aesthetically and proportionally:
During your consultation, Dr. Turkeltaub can assess your unique anatomy, aesthetic goals and lifestyle to determine which, if any, of these complementary procedures may help maximize your surgical outcome. Whether performed individually or in tandem, these options are designed to enhance your confidence and deliver results that are both beautiful and balanced.
Choosing the right breast augmentation surgeon is very important in helping to maximize the likelihood that you will receive excellent care, safe and ethical treatment and results that meet—or even exceed—your breast enhancement goals. Some of the most important factors to consider when making your decision include:
In the end, you should have no reservations about the breast augmentation surgeon you ultimately select, so take your time, do your research and only move forward with treatment when you are entirely comfortable with your choice.
It is likely that you will eventually need to replace your breast implants, as they are not considered “lifetime devices.” Implants can last up to 30 or more years, with silicone implants typically lasting significantly longer than saline implants. Dr. Turkeltaub recommends long-term follow ups with your plastic surgeon to check on the health of your devices and to ensure any potential issues can be addressed efficiently.
Swimming should be avoided after breast augmentation for at least six weeks but this does vary based on individual factors. Dr. Turkeltaub can provide you with a more accurate timeline, based on your individual recovery. You can, however, submerge in a pool at or around four weeks postoperatively.
It is important to protect your scars from the sun for at least six to nine months following surgery. A sunscreen with a SPF of 45 or greater can be applied to the scars when out in the sun even when wearing a bathing suit.
Dr. Turkeltaub is a very experienced plastic surgeon who has performed a large number of breast augmentations in over 30 years of practice. The following are examples of his expertise in this procedure.
Due to social media policies, the following images have been censored. Please click on the button below to view the image.
“One year ago, I made one of the best decisions of my life to have you perform my breast augmentation surgery! I am quite pleased with my result. The boost in confidence I experienced due to the surgery led me to compete in two fitness shows this year. I plan to compete on a national level in November. I want to thank you and your great staff for the professional but very caring experience I had last year.”
For additional helpful information on a variety of issues related to breast augmentation, you can visit Dr. Turkeltaub’s Blog.
Dr. Turkeltaub is also experienced in a variety of other breast surgery procedures, including breast lift, breast reduction, and breast reconstruction.
If you are interested in breast augmentation, please contact Dr. Turkeltaub to schedule a convenient consultation. With two locations, Dr. Turkeltaub’s practice is easily accessible.
We look forward to helping you.